https://doi.org/10.1002/2211-5463.13213
Abstract
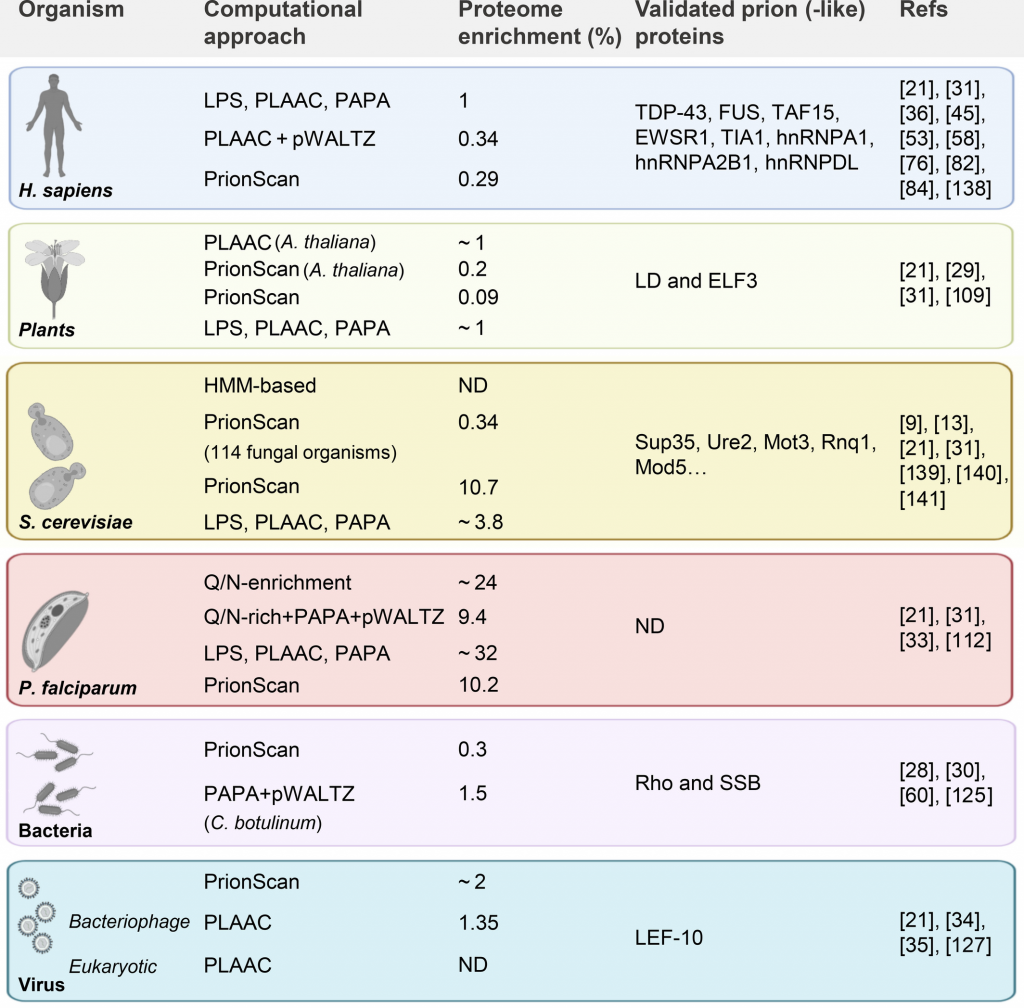
Prions are self-perpetuating proteins able to switch between a soluble state and an aggregated-and-transmissible conformation. These proteinaceous entities have been widely studied in yeast, where they are involved in hereditable phenotypic adaptations. The notion that such proteins could play functional roles and be positively selected by evolution has triggered the development of computational tools to identify prion-like proteins in different kingdoms of life. These algorithms have succeeded in screening multiple proteomes, allowing the identification of prion-like proteins in a diversity of unrelated organisms, evidencing that the prion phenomenon is well conserved among species. Interestingly enough, prion-like proteins are not only connected with the formation of functional membraneless protein–nucleic acid coacervates, but are also linked to human diseases. This review addresses state-of-the-art computational approaches to identify prion-like proteins, describes proteome-wide analysis efforts, discusses these unique proteins’ functional role, and illustrates recently validated examples in different domains of life.
