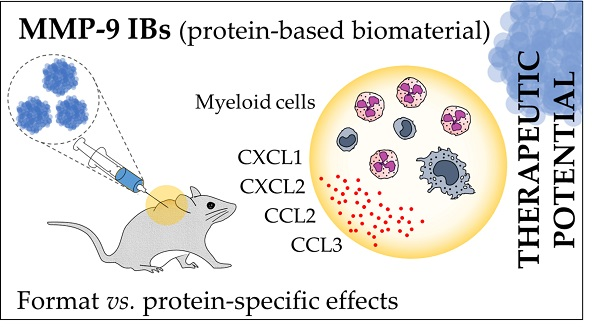
Bacterial inclusion bodies (IBs) are protein-based nanoparticles of a few hundred nanometers formed during recombinant protein production processes in different bacterial hosts. IBs contain active protein in a mechanically stable nanostructured format that has been broadly characterized, showing promising potential in different fields such as tissue engineering, protein replacement therapies, cancer, and biotechnology. For immunomodulatory purposes, however, the interference of the format immunogenic properties—intrinsic to IBs—with the specific effects of the therapeutic protein is still an uncovered gap. For that, active and inactive forms of the catalytic domain of a matrix metalloproteinase-9 (MMP-9 and mutMMP-9, respectively) have been produced as IBs and compared with the soluble form for dermal inflammatory effects in mmp9 knock-out mice. After protein injections in air-pouches in the mouse model, MMP-9 IBs induce local neutrophil recruitment and increase pro-inflammatory chemokine levels, lasting for at least two days, whereas the effects triggered by the soluble MMP-9 format fade out after 3 h. Interestingly, the IB intrinsic effects (mutMMP-9 IBs) do not last more than 24 h. Therefore, it may be concluded that IBs could be used for the delivery of therapeutic proteins, such as immunomodulating proteins while preserving their stability in the specific tissue and without triggering important unspecific inflammatory responses due to the protein format.